
Electrical Conductivity (EC) reflects the concentration of soluble salts in soil or irrigation water. High EC indicates salinity stress that hampers plant growth, while moderate levels help assess fertilizer availability. Monitoring EC is a quick and reliable way to optimize nutrient management and prevent yield losses.
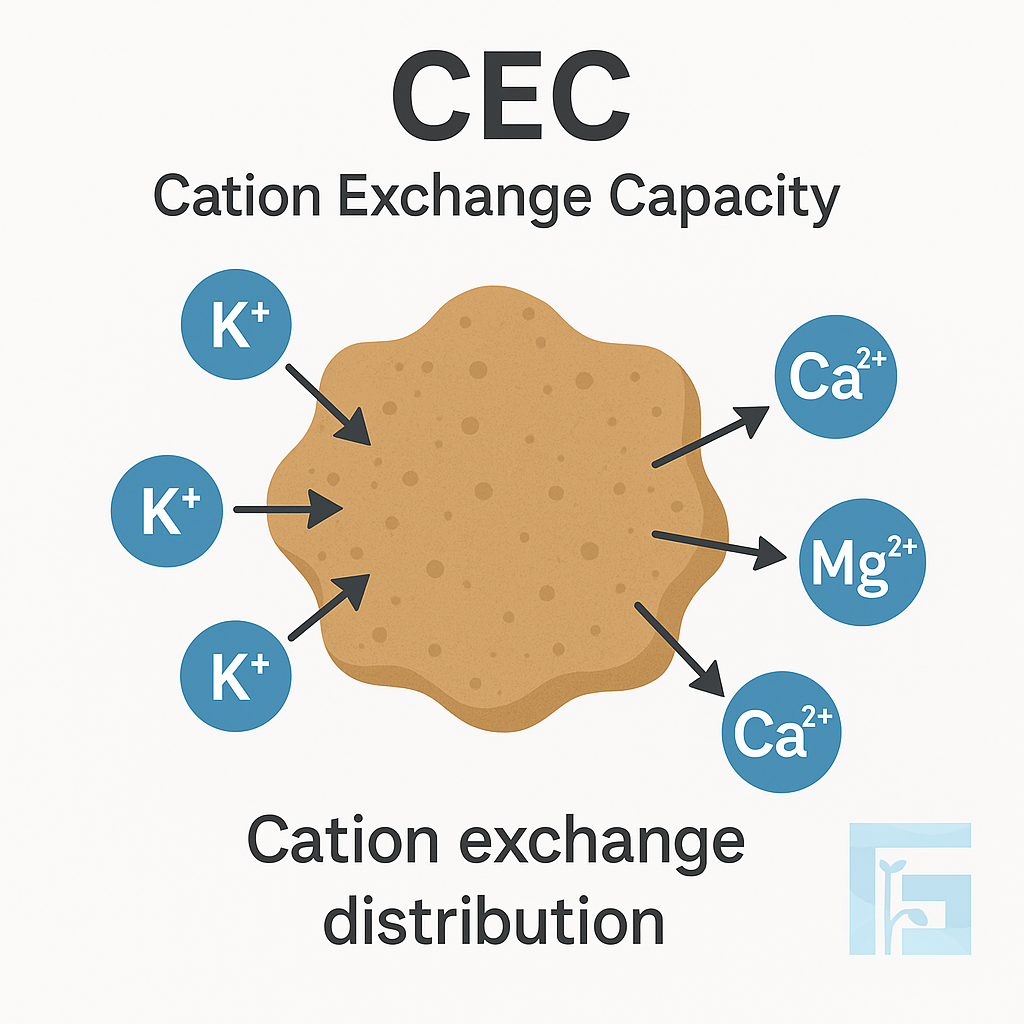
Cation Exchange Capacity (CEC) measures the soil’s ability to retain and supply positively charged nutrients such as potassium (K⁺), calcium (Ca²⁺), and magnesium (Mg²⁺). Soils with higher CEC act like nutrient reservoirs, supporting better fertility and buffering against nutrient losses. Understanding CEC helps farmers choose the right fertilizers and manage soil health more sustainably.
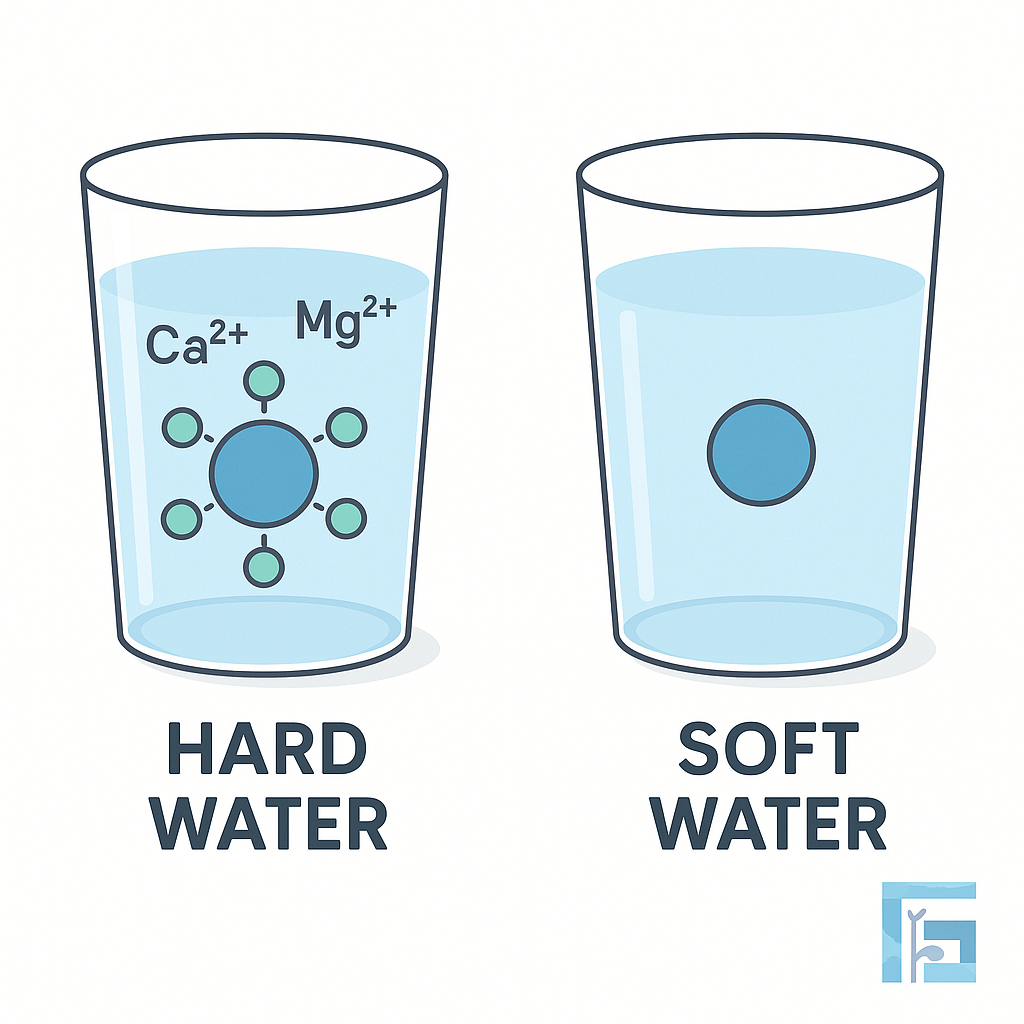
Water hardness is caused by dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions. In agriculture, hard water can reduce the effectiveness of pesticides and fertilizers by forming insoluble precipitates or binding with active ingredients. Understanding and managing water hardness is essential for ensuring spray solution stability and maximizing crop protection efficacy.
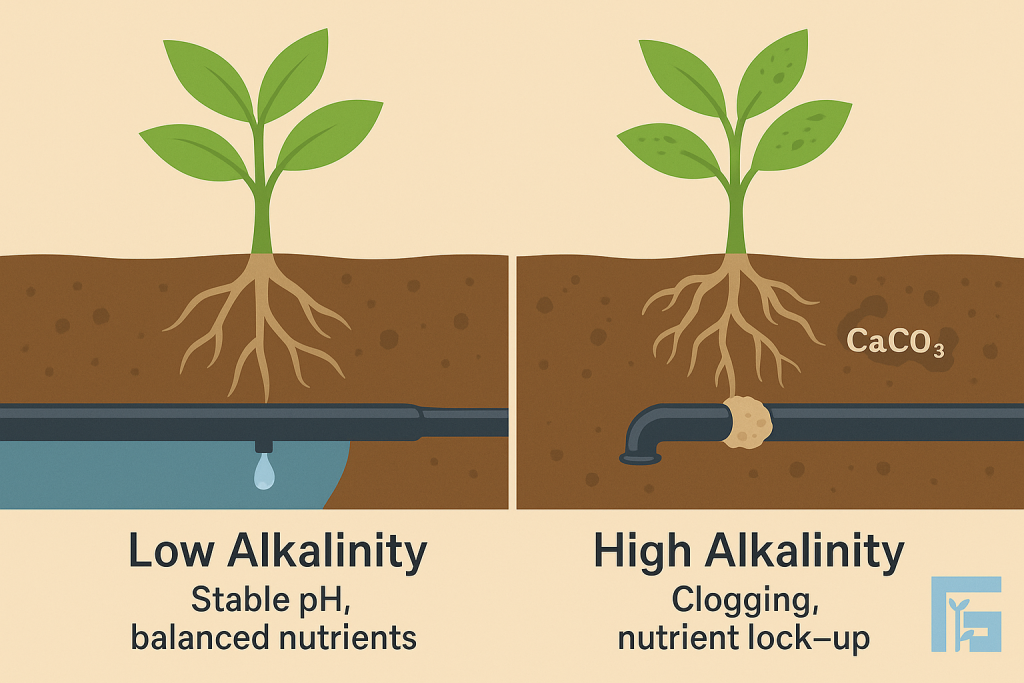
Alkalinity in irrigation water, mainly from bicarbonates (HCO₃⁻) and carbonates (CO₃²⁻), affects pH stability and nutrient availability. High alkalinity can cause precipitation of calcium and magnesium, leading to clogged irrigation systems and reduced fertilizer efficiency. Managing alkalinity is critical to maintain soil balance and crop performance.

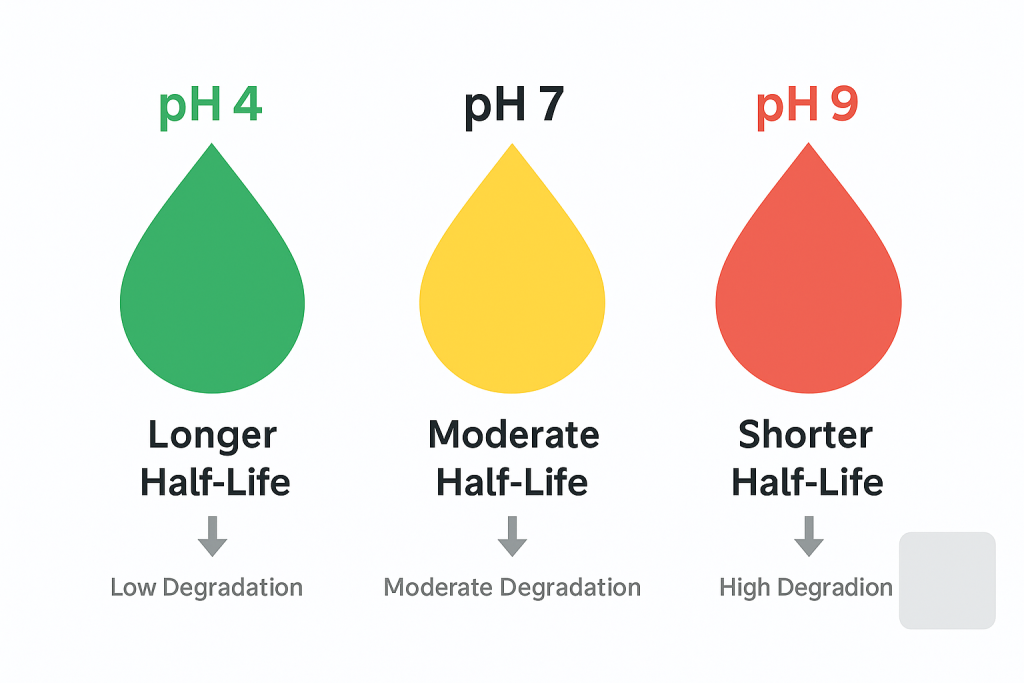
Pesticide stability is heavily influenced by spray solution pH; for example, the fungicide fluazinam remains stable at acidic pH (pH 4) but degrades rapidly at neutral to alkaline pH (pH 7–9). This shift in stability affects half-life and field efficacy. Adjusting water pH to appropriate levels is essential to preserve pesticide performance and consistency.

Redox Potential (Eh) indicates the tendency of soil to either gain or lose electrons. Under high Eh (oxidizing conditions), nutrients like nitrate (NO₃⁻) and sulfate (SO₄²⁻) dominate, while low Eh (reducing conditions) favors forms like ammonium (NH₄⁺), iron (Fe²⁺), and manganese (Mn²⁺). Monitoring Eh helps predict nutrient availability, metal toxicity, and soil microbial activity in flooded or waterlogged soils.

Micronutrients such as zinc (Zn), iron (Fe), manganese (Mn), and copper (Cu) are required in small amounts, but they play vital roles in enzyme activation, photosynthesis, and plant defense. Deficiency of these trace elements leads to chlorosis, stunted growth, and reduced yields, making balanced micronutrient management essential for sustainable agriculture.
Surfactants reduce surface tension, allowing pesticide droplets to spread evenly on leaf surfaces rather than remaining as beads. This enhanced coverage improves penetration of active ingredients through the waxy cuticle, leading to greater biological efficacy. By optimizing distribution, surfactants help reduce chemical waste and environmental impact.

Buffering capacity is the soil’s ability to resist changes in pH when acids or bases are added. Soils with high organic matter and clay content have stronger buffering power, protecting crops from sudden nutrient imbalances. Understanding buffering capacity is essential for precise lime or fertilizer management.
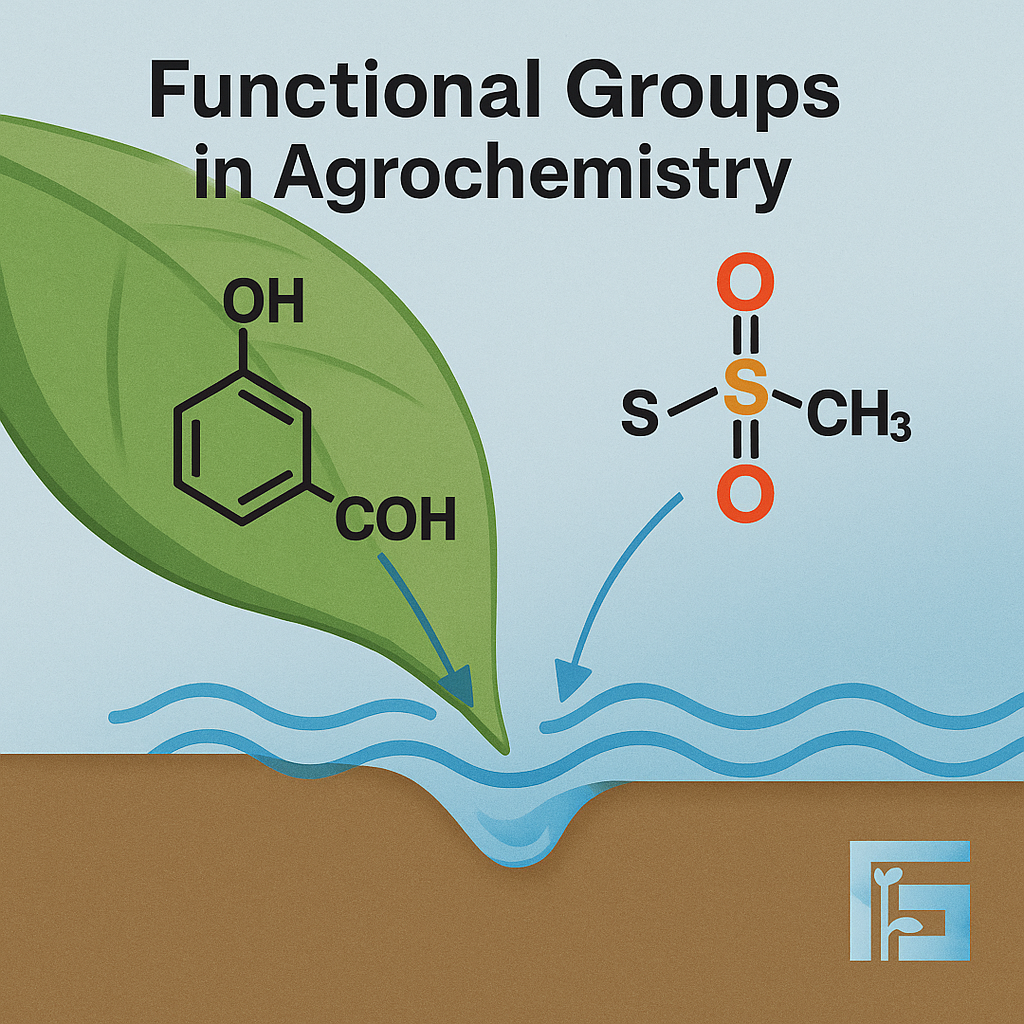
Functional groups such as hydroxyl, carboxyl, and sulfonyl determine agrochemical solubility, stability, and interaction with plant and soil matrices. Sulfur-based groups play pivotal roles in crop protection agents like dithiocarbamates and sulfonylureas, enabling targeted action and molecular stability. Rational design of these functional moieties is key to crafting efficient and environmentally compatible formulations.
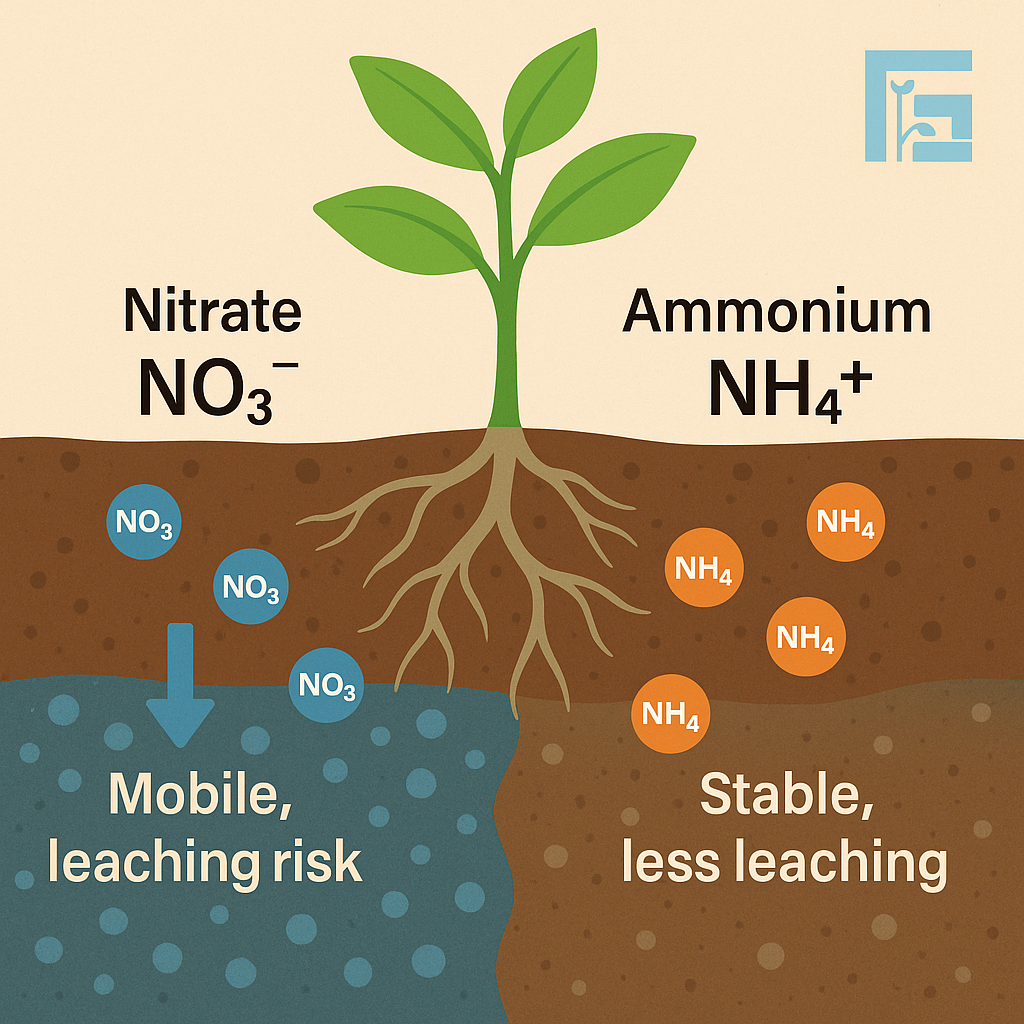
Nitrogen in soil primarily exists as nitrate (NO₃⁻) and ammonium (NH₄⁺). Nitrate is mobile, easily leached, and quickly available to plants but prone to losses. Ammonium, on the other hand, binds to soil particles, releases slowly, and reduces leaching risks. Balancing these two forms is crucial for efficient fertilization and sustainable crop yields.

Chelated micronutrients remain soluble and stable in soil, protecting essential metals such as Fe, Zn, and Mn from precipitation or fixation. This stability enhances their bioavailability and facilitates easier transport across plant membranes. In contrast, inorganic salts often undergo rapid reactions, limiting nutrient uptake efficiency.
Nanotechnology: Smarter Delivery Systems for Fungicides
Nanoparticles can encapsulate fungicides, protecting them from degradation and enabling controlled release at the infection site. This targeted delivery improves efficacy against fungal pathogens while reducing overall chemical usage. Such innovations promise sustainable crop protection with minimized environmental impact.
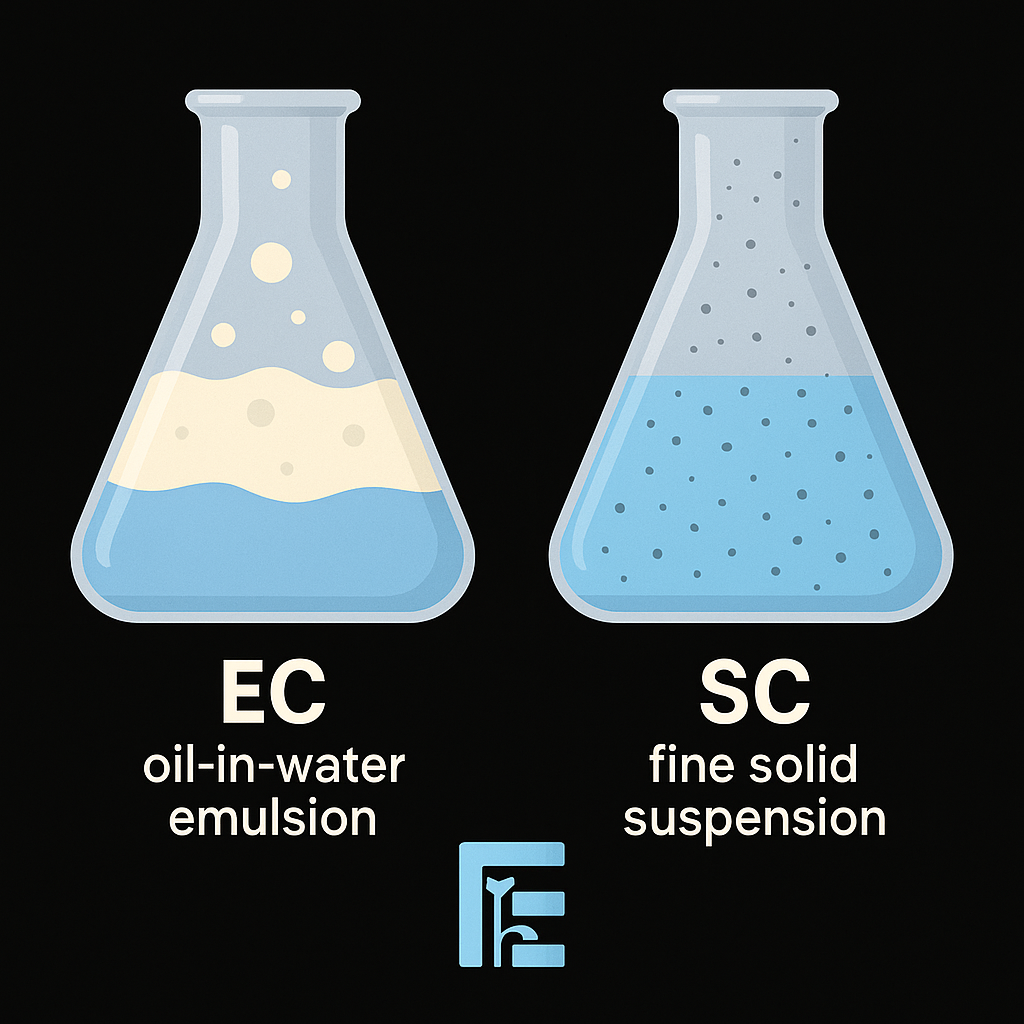
Emulsifiable concentrates (EC) dissolve the active ingredient in organic solvents, forming emulsions upon dilution in water. In contrast, suspension concentrates (SC) disperse fine solid particles in water with stabilizers, improving safety and reducing volatile emissions. The choice between EC and SC affects stability, application efficiency, and environmental footprint.
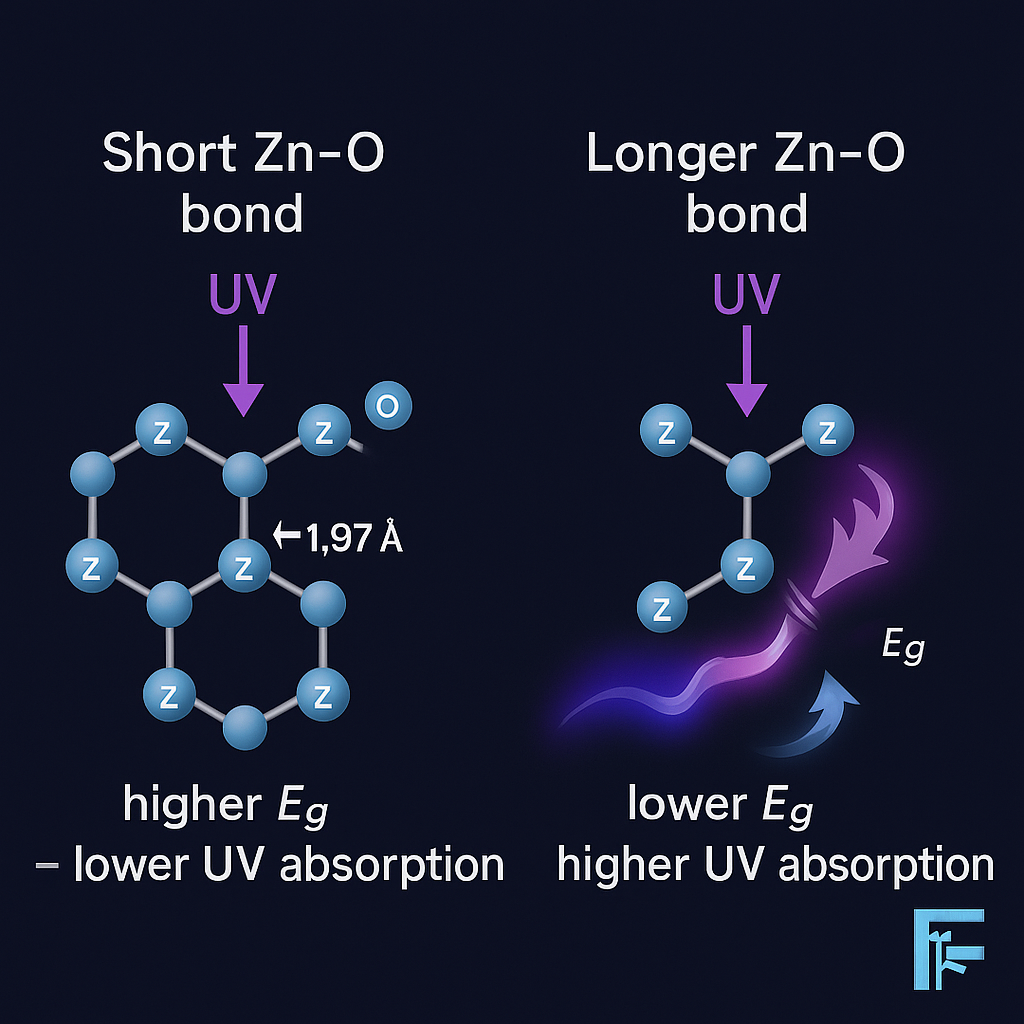
Variations in Zn–O bond length, influenced by laser irradiation or doping, can modulate the bandgap and shift UV-absorption peaks of ZnO nanoparticles. A slight increase in bond length tends to lower the bandgap, enhancing UV absorption in the 250–400 nm range. This fine-tuning offers valuable control over optical properties for UV-filtering applications in agrochemistry.
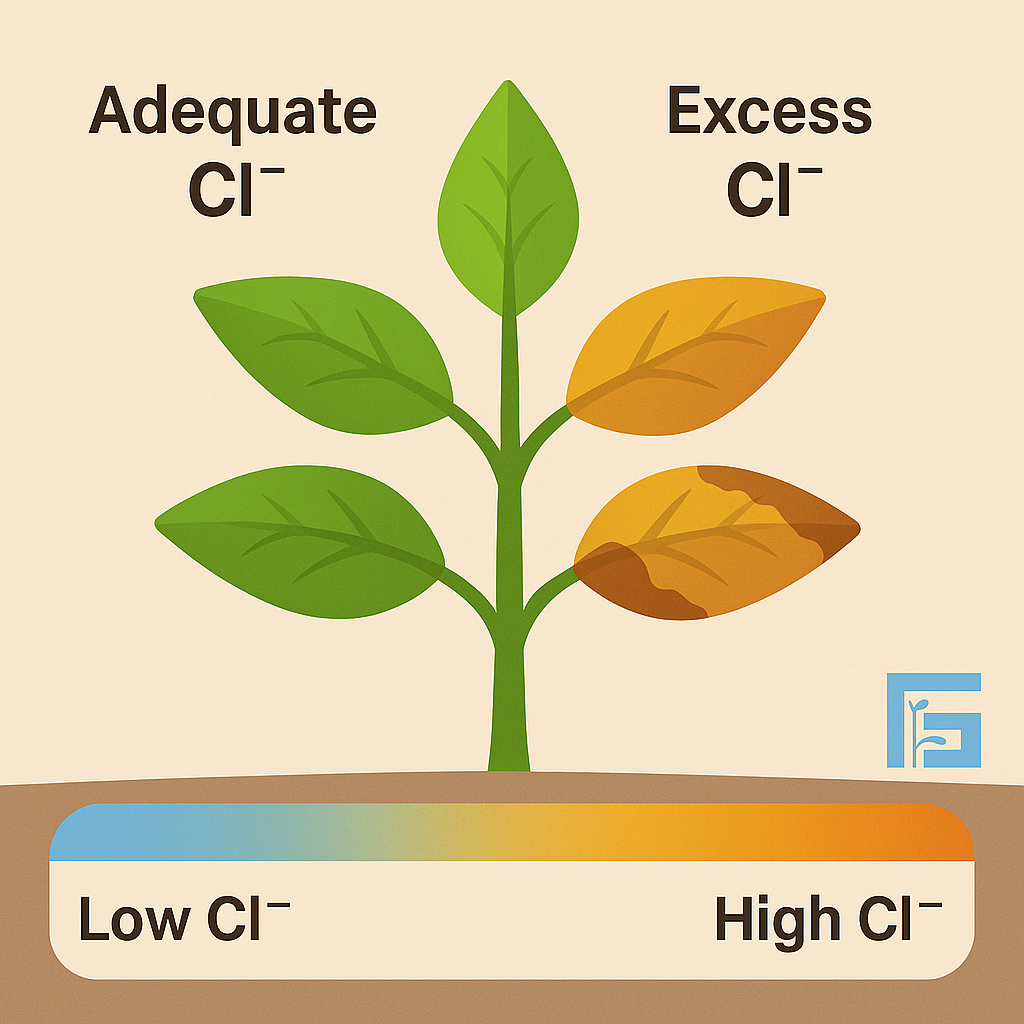
Chloride (Cl⁻) is an essential micronutrient for plants, supporting photosynthesis and osmotic balance. However, excessive chloride in irrigation water accumulates in leaves, causing tip burn, reduced photosynthetic activity, and yield decline. Sensitive crops like grapes and citrus are especially at risk.

The Sodium Adsorption Ratio (SAR) reflects the proportion of sodium relative to calcium and magnesium in irrigation water. Elevated SAR levels cause soil dispersion, reduce water infiltration, and limit root aeration. Proper monitoring and management of SAR safeguard soil structure and crop performance.
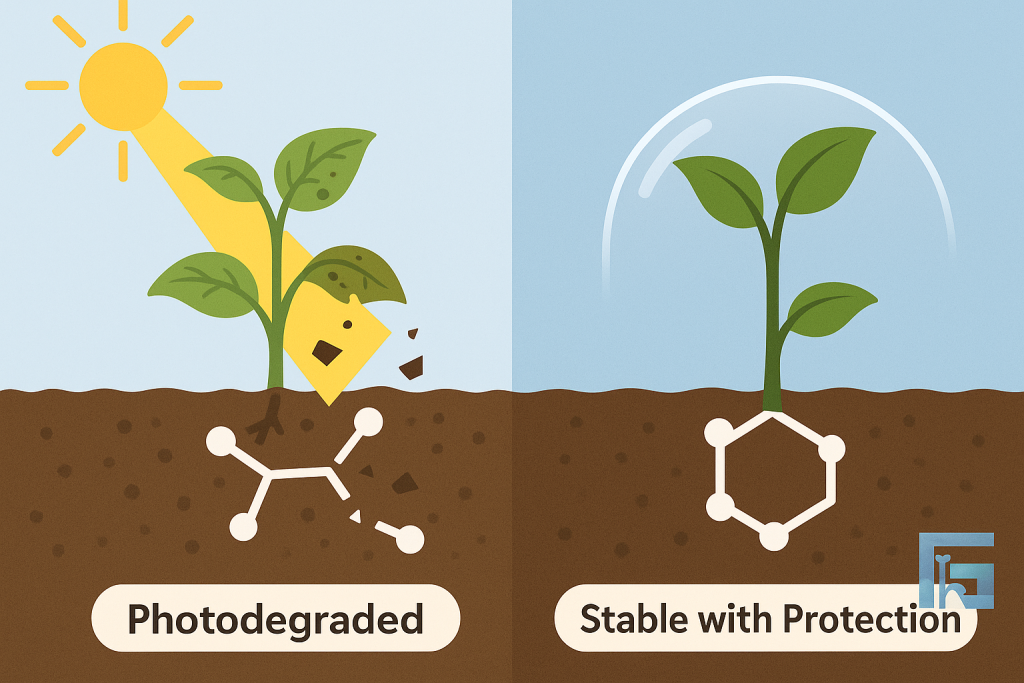
Photodegradation is the breakdown of pesticide molecules under sunlight, often leading to rapid loss of field efficacy. UV light excites chemical bonds, causing them to split and form less active or inactive products. Understanding photodegradation helps in choosing stable formulations, applying sprays at the right time, and developing UV-protective additives for longer-lasting protection.

In calcareous soils with high pH and lime content, iron (Fe) becomes insoluble and unavailable to plants. This results in interveinal chlorosis, where leaves turn yellow while veins remain green. Iron deficiency is one of the most widespread nutrient disorders in crops grown on alkaline soils.
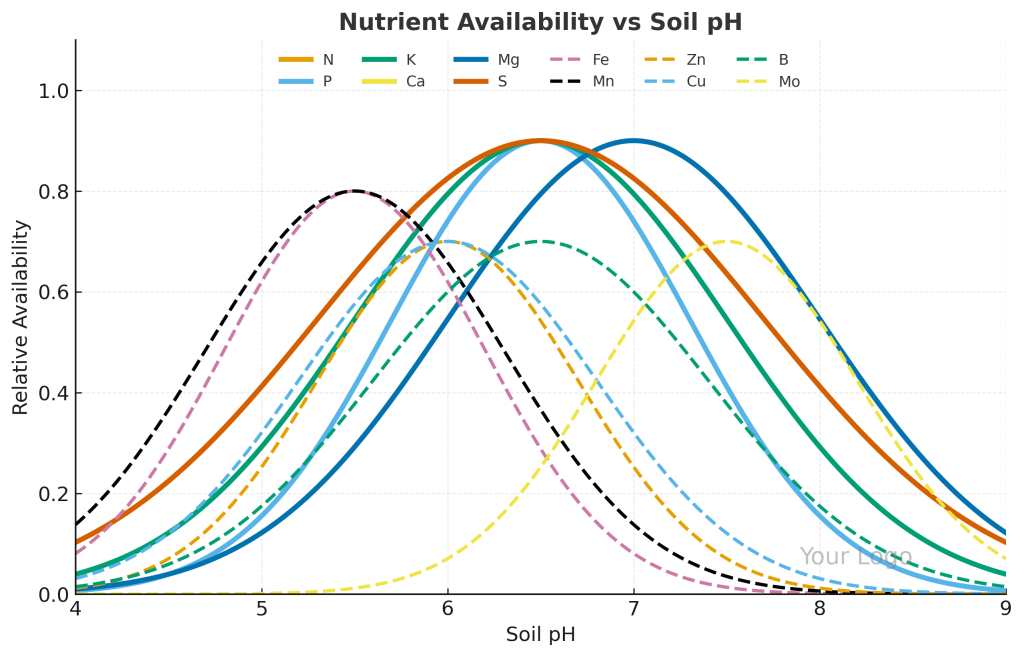
Soil pH strongly controls nutrient availability and plant health. In acidic soils, phosphorus and molybdenum become deficient, while toxic levels of aluminum and manganese may appear. In alkaline soils, iron, zinc, and manganese deficiencies are common, leading to chlorosis and reduced growth.
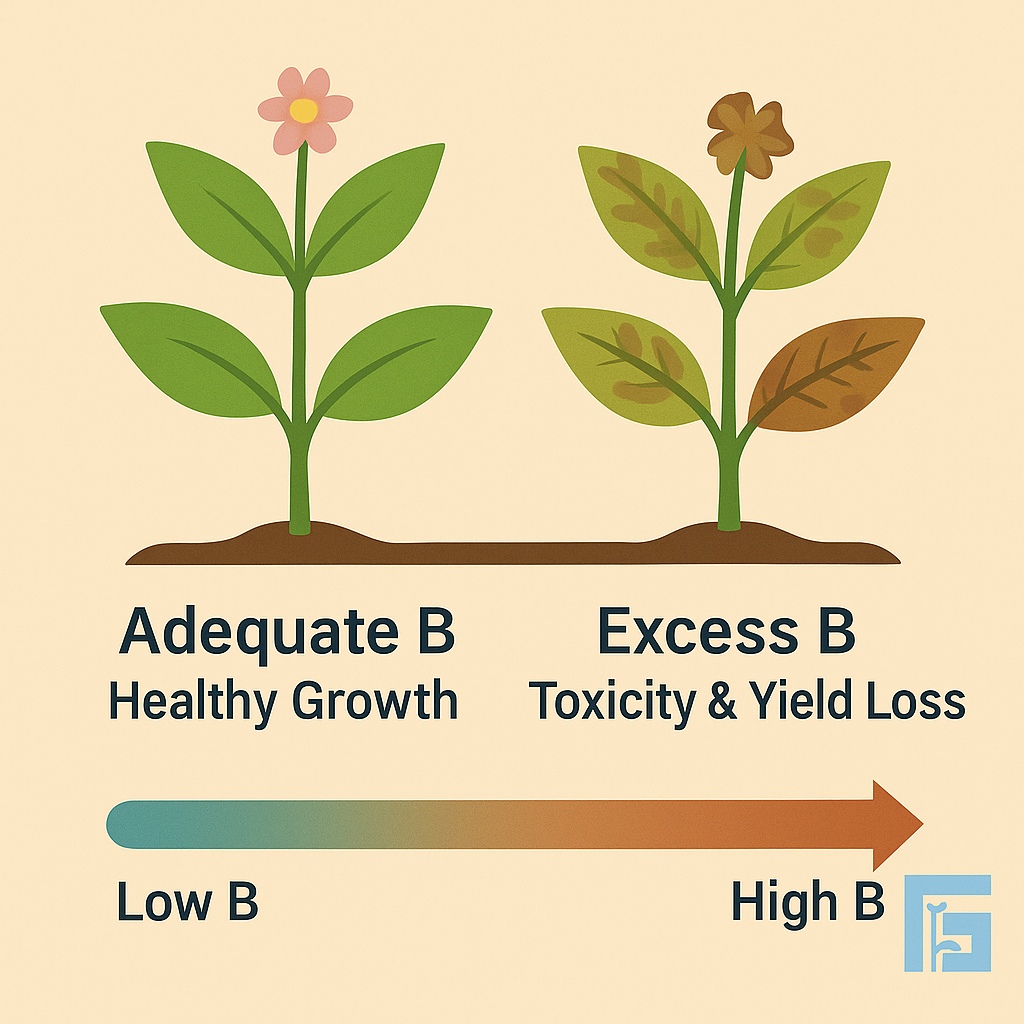
Boron (B) is essential for cell wall development, sugar transport, and reproductive growth in plants. Yet, when irrigation water contains high boron, it quickly turns toxic, causing leaf burn, chlorosis, and reduced yields. Careful monitoring is crucial, especially in arid and semi-arid regions.
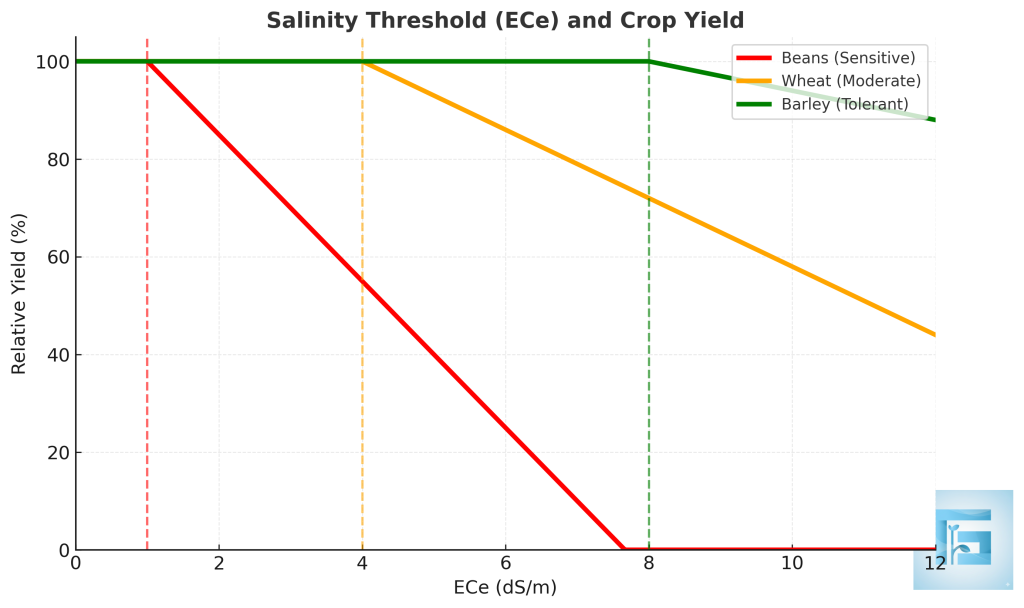
Each crop has a salinity threshold (ECe, dS/m) below which yield remains unaffected. Beyond this threshold, yield decreases proportionally with increasing soil salinity. Salt-sensitive crops like beans decline quickly, while tolerant crops like barley sustain production under higher ECe values.
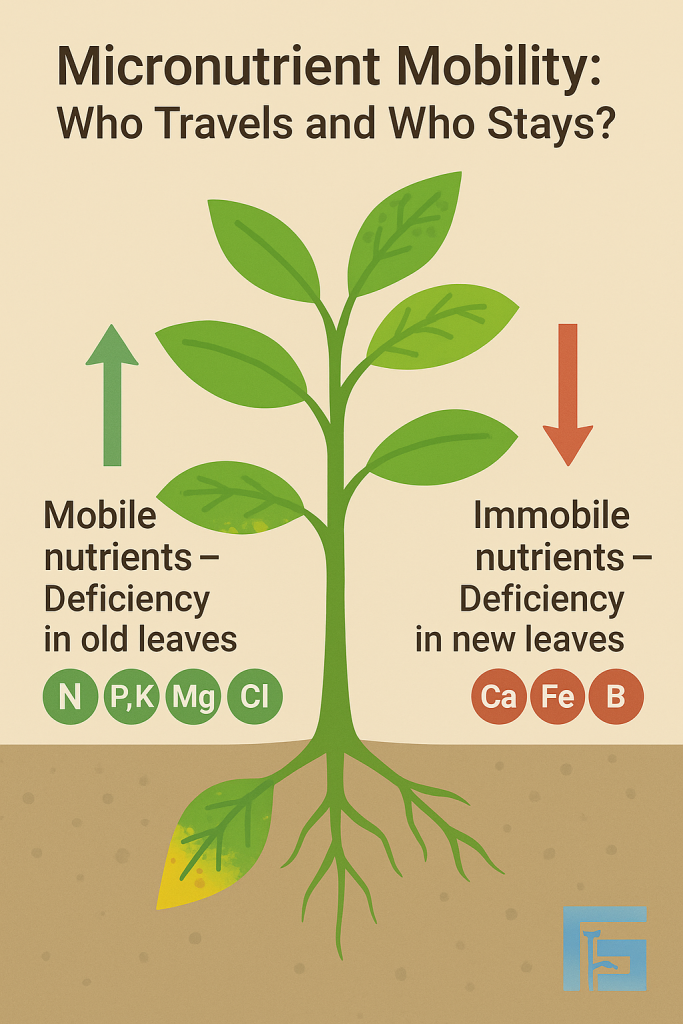
Nutrient mobility in plants determines where deficiency symptoms first appear. Mobile nutrients (like N, P, K, Mg, Cl) move to young leaves, so deficiencies show up in older leaves. Immobile nutrients (like Ca, Fe, B) remain in young tissues, making deficiency symptoms visible in new growth.

Nutrient antagonism occurs when an excess of one nutrient reduces the uptake of another. For example, high potassium can suppress magnesium and calcium absorption, while excessive phosphorus can limit zinc and iron availability. Recognizing these interactions is key to balanced fertilization.

Electrical Conductivity (EC) measures the ability of water to conduct electricity, which depends on dissolved salts. Total Dissolved Solids (TDS) expresses the same salinity in milligrams per liter (mg/L). TDS is typically estimated from EC using a conversion factor, making EC a faster, indirect indicator of water salinity.

Total Dissolved Solids (TDS) represent the sum of inorganic salts, minerals, and small organic matter dissolved in water. High TDS levels can reduce water quality, affect nutrient uptake, and cause salinity stress in crops. Monitoring TDS helps farmers ensure safe irrigation practices and protect plant health.

Sodic soils, dominated by sodium (Na⁺), suffer from poor structure, dispersion, and low infiltration. Applying gypsum (CaSO₄·2H₂O) replaces sodium with calcium, flocculating soil particles and restoring porosity. This amendment is a practical solution to reclaim sodic soils for sustainable farming.
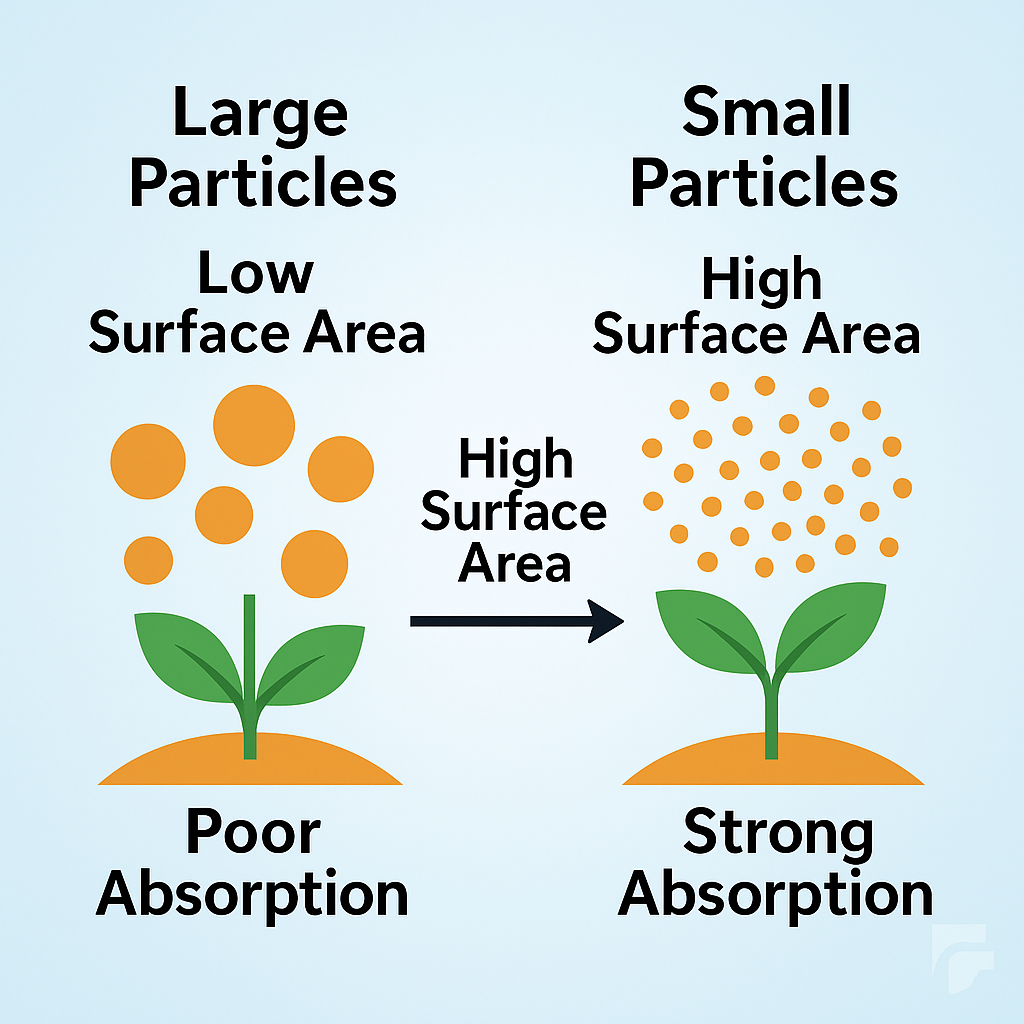
Reducing particle size increases the surface area of active ingredients, enhancing their dissolution rate and availability in spray solutions. Smaller particles penetrate plant surfaces more efficiently and distribute more uniformly on leaves or soil. Micronization and nanonization technologies now significantly improve efficacy while lowering total chemical use.
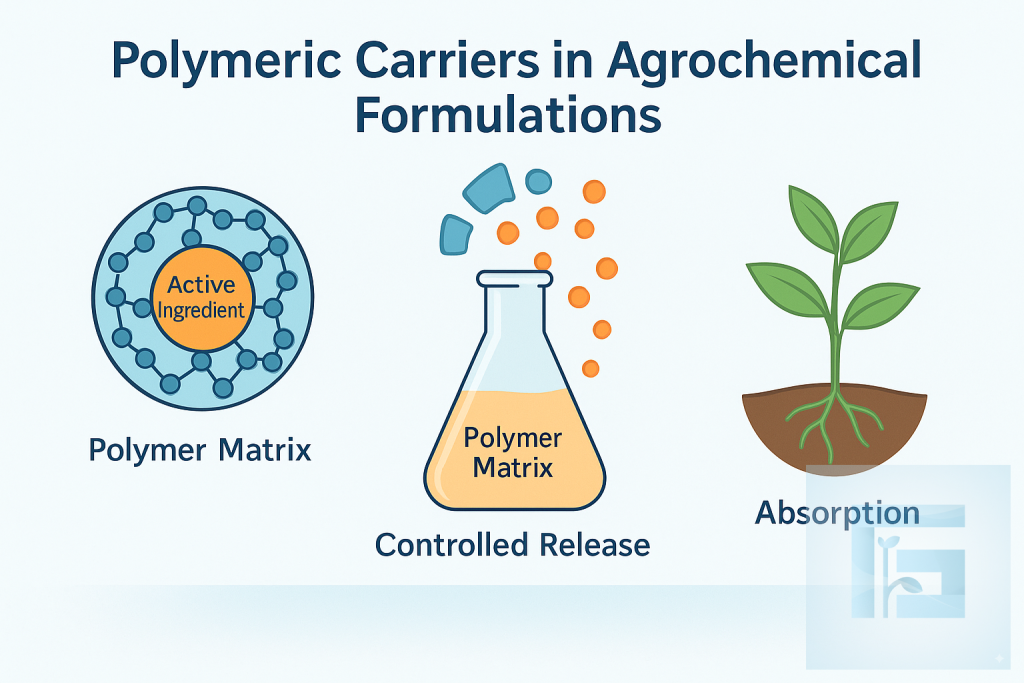
Polymeric carriers encapsulate active ingredients, protecting them from premature degradation and enabling controlled release in soil or on plant surfaces. This controlled delivery improves bioavailability, reduces application frequency, and enhances environmental safety. Advances in biodegradable polymers now offer highly efficient and sustainable agrochemical systems.
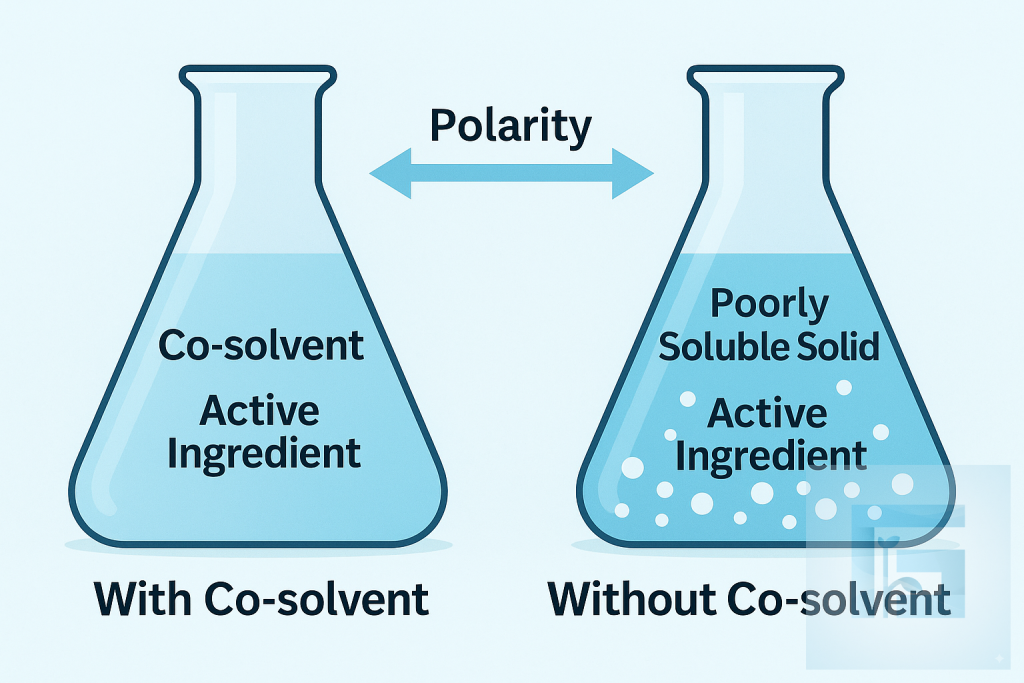
Co-solvents improve the solubility of poorly water-soluble active ingredients by modifying the polarity and hydrogen-bonding properties of the formulation. Their presence enables stable concentrates, prevents crystallization, and enhances uniform dispersion in spray solutions. Selecting the right co-solvent is critical for achieving optimal efficacy, storage stability, and field performance.
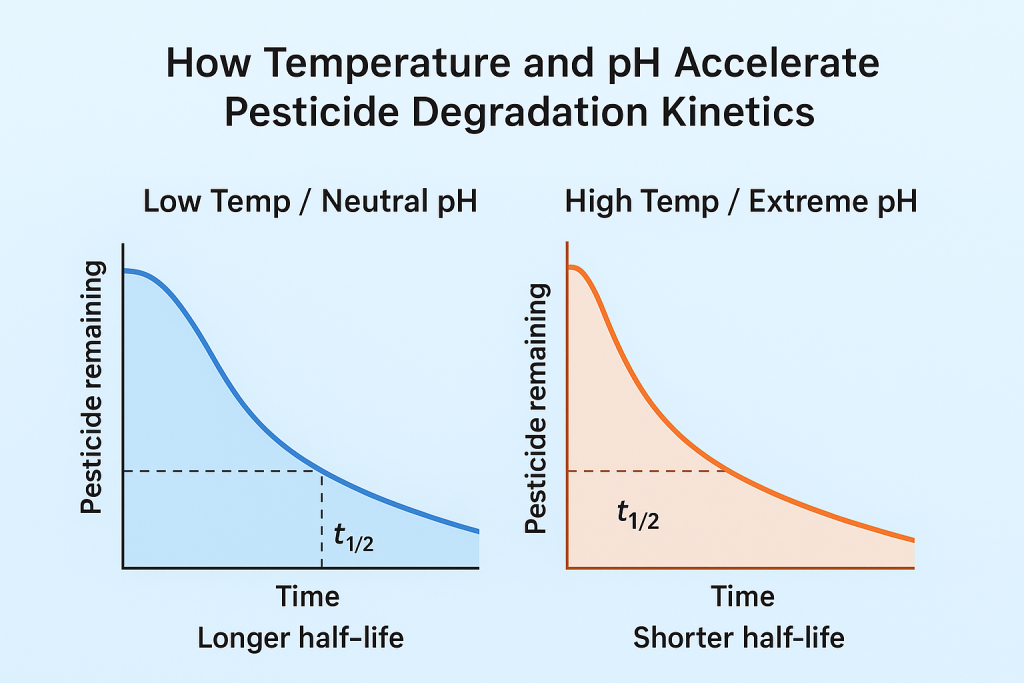
Temperature and pH significantly influence the hydrolysis and breakdown rate of many pesticides, often shortening their field half-life. Higher temperatures increase molecular motion, accelerating degradation, while extreme pH levels promote hydrolysis of sensitive active ingredients. Understanding these kinetic shifts helps optimize spray timing and preserve efficacy under variable environmental conditions.
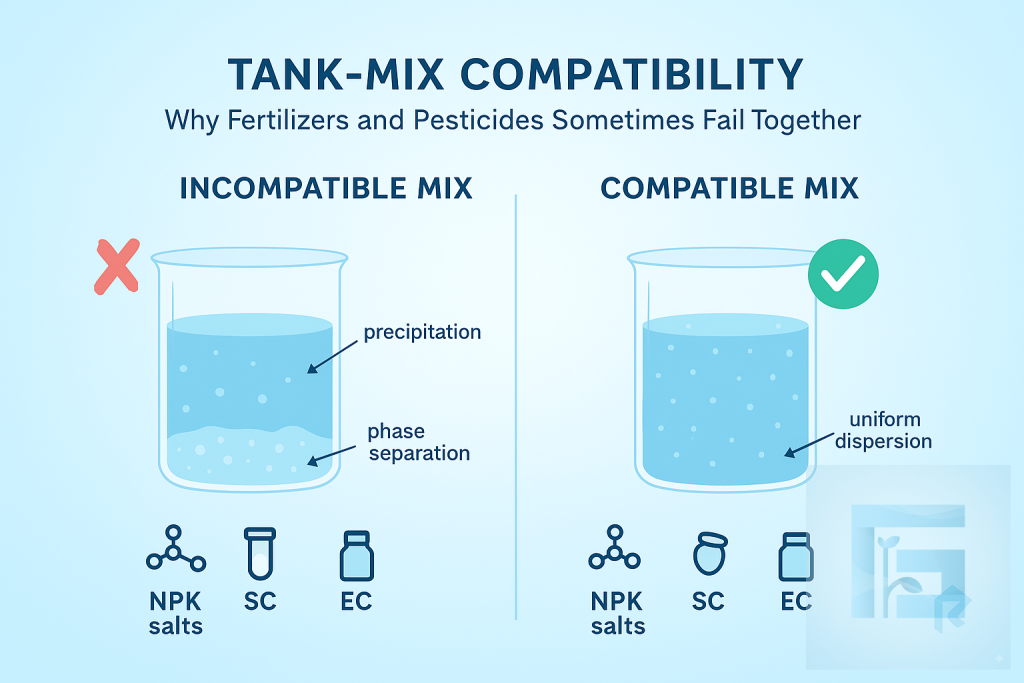
Chemical incompatibilities during tank-mixing can lead to precipitation, phase separation, or degradation of active ingredients. Fertilizer salts may alter pH or ionic strength, destabilizing pesticide formulations such as SCs or ECs. Understanding compatibility charts and conducting a simple jar test are essential steps to avoid reduced efficacy and spraying failures.
Oils and organosilicone adjuvants increase droplet spreading, cuticular penetration, and overall deposition efficiency far beyond conventional surfactants. Organosilicones reduce surface tension to exceptionally low levels, enabling rapid stomatal infiltration and enhanced active ingredient uptake. These advanced adjuvants can significantly reduce spray volume while improving bioefficacy and uniformity.


